Keywords: Mg-based alloy, Biodegradable metallic material, Intermetallic compound, Degradation behavior
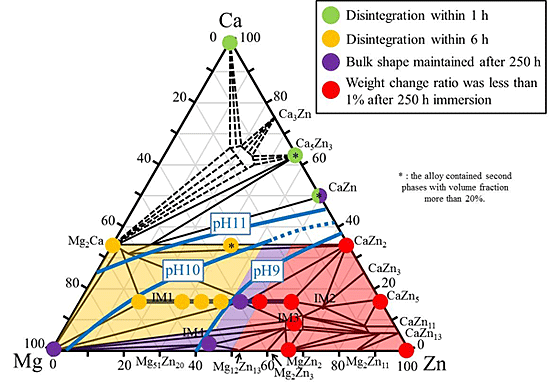
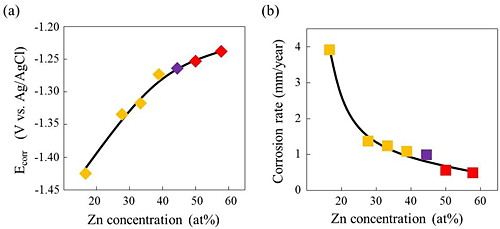
With the goal of developing new biodegradable implant materials, we have investigated the degradation behavior of (Ca, Mg)-based intermetallic compounds. The degradation behavior of the compounds within the Ca-Mg-Zn system was roughly classified into four groups as shown in Fig. 1. Their degradation behaviors were strongly influenced by the compositions of the compounds. For example, the Ca3MgxZn(15 - x) compound exhibited a large solubility region with varying the Mg/Zn ratio, and the corrosion behavior was largely varied by the composition, as was obviously understood in Fig. 2. Indeed, the Ca3Mg12Zn3 phase alloy with the lowest Zn content was rapidly broken apart within 6 h of immersion. Alternatively, the Ca3Mg4.6Zn10.4 phase alloy with the highest Zn content retained the bulk shape even after 250 h of immersion as shown in Fig. 3. These varying degradation behaviors were ascribed to the difference in the formability of Zn oxide as a protective layer against corrosion on the specimen surfaces, depending on the Zn content. The gained results suggest that there is a feasibility on developing new biodegradable materials based on intermetallic compounds in which the degradation rate can be controlled by their compositions. The fabrication of the 3-dimensional porous biodegradable materials has been considered as a future work by using these (Ca, Mg)-based intermetallic compounds.