State changes of intermetallic compounds during the high temperature and short
time solution treatments were investigated for an AC4CH aluminum alloy. The
solution treatments were carried out using the newly developed high-frequency
induction heating apparatus equipped with the frequency of 2MHz and the power of
10kW. Microscopic observations using the optical microscope and the electron probe
micro analyzer revealed the following remarks. Mg atoms in the as-cast specimen
were segregated in the intermetallic compounds such as Mg2Si-phase and ![]() -phases. The configuration of
-phases. The configuration of ![]() -phase was classified into the Chinese script-like and the plate-like shape. The Mg2Si phase was dissolved easily by heating up to 560
-phase was classified into the Chinese script-like and the plate-like shape. The Mg2Si phase was dissolved easily by heating up to 560![]() . The solution treatment at 560
. The solution treatment at 560![]() for 3 minutes brought about not only the dissolution of the Chinese script-like
for 3 minutes brought about not only the dissolution of the Chinese script-like ![]() -phase but also the transformation of the plate-like
-phase but also the transformation of the plate-like ![]() -phase to the
-phase to the ![]() -AlFeSi phase. These state changes of the Mg2Si phase and
-AlFeSi phase. These state changes of the Mg2Si phase and ![]() -phases resulted in the significant increase of solute Mg and Si atoms in the primary
-phases resulted in the significant increase of solute Mg and Si atoms in the primary ![]() -aluminum phase. It was also confirmed that the solution time required for the dissolution or the transformation of
-aluminum phase. It was also confirmed that the solution time required for the dissolution or the transformation of ![]() -phases becomes longer with coarsening of
-phases becomes longer with coarsening of ![]() -phases.
-phases.
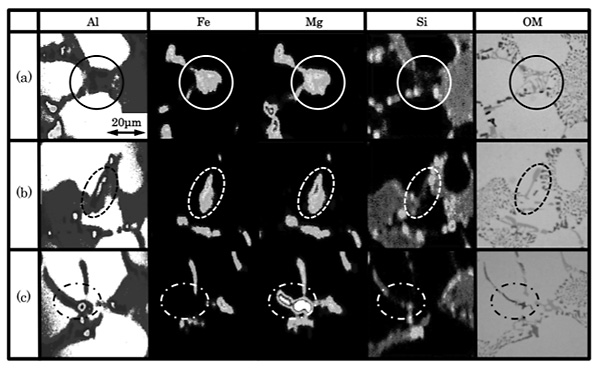
Element mappings of the as-cast specimen are shown in Fig. 2 together with the optical micrographs at the same position. Fig. 2 reveals that microstructure of the as-cast specimen consists of both the primary ![]() -aluminum and the eutectic region with network shape. Si atoms segregate in the eutectic region, whereas coincidence concentration of Fe and Mg atoms is clearly confirmed in the specific position of eutectic region.
-aluminum and the eutectic region with network shape. Si atoms segregate in the eutectic region, whereas coincidence concentration of Fe and Mg atoms is clearly confirmed in the specific position of eutectic region.
Fig. 3 shows the EPMA mapping and the optical micrograph of intermetallic compounds. On the basis of the geometrical configuration and the distribution of Fe, Mg, and Si atoms, intermetallic compounds are classified into three types. Chinese script-like and plate-like compounds seem to be an Al-Fe-Mg-Si intermetallic compound, ![]() -phase. In addition to the above, elemental mapping shows that another plate-like compound is to be an Mg2Si phase.
-phase. In addition to the above, elemental mapping shows that another plate-like compound is to be an Mg2Si phase.
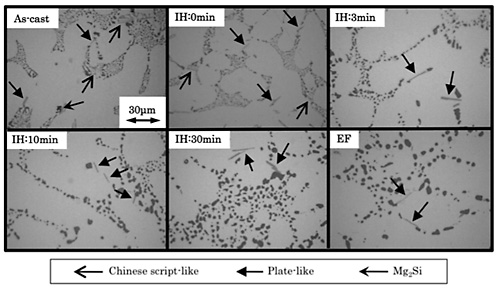
Changes in intermetallic compounds with the high temperature and short time solution treatment are summarized in Fig. 5, together with microstructure treated under the standard solution condition (at 535![]() for 480 minutes) using the conventional electric furnace, described by "EF". Mg2Si phase dissolved at the solution treatment time of 0 minute, suggesting the lower heat stability of Mg2Si phase.
for 480 minutes) using the conventional electric furnace, described by "EF". Mg2Si phase dissolved at the solution treatment time of 0 minute, suggesting the lower heat stability of Mg2Si phase. ![]() -phase with Chinese script-like shape disappeared at the solution time of 3 minutes, whereas the intermetallic compounds with plate-like shape was still observed at the longest solution time of 30 minutes. Quantitative analysis using EPMA suggested that the plate-like compound was to be
-phase with Chinese script-like shape disappeared at the solution time of 3 minutes, whereas the intermetallic compounds with plate-like shape was still observed at the longest solution time of 30 minutes. Quantitative analysis using EPMA suggested that the plate-like compound was to be ![]() -AlFeSi phase. Therefore, the solution treatment seems to prompt the transformation from plate-like
-AlFeSi phase. Therefore, the solution treatment seems to prompt the transformation from plate-like ![]() -phase to
-phase to ![]() -AlFeSi phase.
-AlFeSi phase.
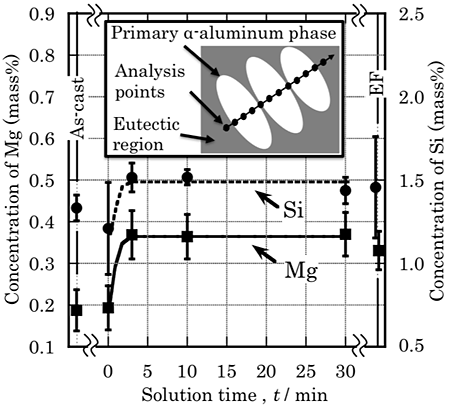
Fig. 6 shows the solution time dependence of the Mg and Si concentrations in the primary ![]() -aluminum. Mg and Si concentration of the as-cast specimen and that at the solution time of 0 minute are relatively low, whereas high concentrations of Mg and Si atoms are measured at the solution time more than 3 minutes. The Mg concentration, about 0.37 mass%, is equivalent to the addition amount of Mg in the sample, on the other hand concentration of Si, about 1.5 mass%, is the value that is almost the maximum solubility limit. These solution time dependences of Mg and Si concentrations in the primary
-aluminum. Mg and Si concentration of the as-cast specimen and that at the solution time of 0 minute are relatively low, whereas high concentrations of Mg and Si atoms are measured at the solution time more than 3 minutes. The Mg concentration, about 0.37 mass%, is equivalent to the addition amount of Mg in the sample, on the other hand concentration of Si, about 1.5 mass%, is the value that is almost the maximum solubility limit. These solution time dependences of Mg and Si concentrations in the primary ![]() -aluminum phase are good agreement with the state changes of intermetallic compounds shown in Fig. 5.
-aluminum phase are good agreement with the state changes of intermetallic compounds shown in Fig. 5.
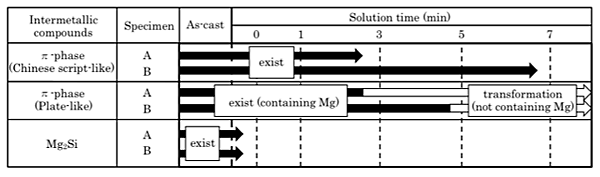
Fig. 9 shows the effect of intermetallic compound size on the state changes of itself. Compared with the specimen A, there are coarse intermetallic compounds such as Mg2Si and ![]() -phases in the specimen B. As shown in Fig. 9, it is clearly shown that the larger
-phases in the specimen B. As shown in Fig. 9, it is clearly shown that the larger ![]() -phase needs the longer solution treatment time. In contrast, the size of Mg2Si phase does not affect the solution treatment time required for state change of itself.
-phase needs the longer solution treatment time. In contrast, the size of Mg2Si phase does not affect the solution treatment time required for state change of itself.