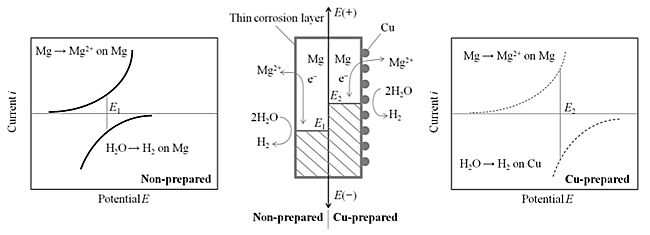
Despite the superior properties of Mg alloys such as high strength, light weight and rich natural resources, weak corrosion resistance limits their practical application. Mg alloys are also easily degraded in the plating process which is necessary to form corrosion protection coating. This paper describes the pretreatment process of AZ31 Mg alloy prior to the electroplating of Cu as an electro-conducting layer. The plating process containing degreasing, pickling, activation, zincating, and Cu electrodeposition. In the activation process, a small amount of Cu2+ ions was added to the activation bath to form fine Cu pre-deposits at high density on the substrate surface accompanying Mg dissolution (Cu pretreatment). These Cu pre-deposits acted as nucleation seeds for Zn deposition in the following zincate process and provided an uniform and dense Zn layer almost completely covering the substrate, as schematically presented in Fig. 1. Efficient charge transfer via Cu pre-deposits to Zn2+ ions also shortens the duration of substitution reaction of Mg dissolution and Zn deposition, resulting in suppression of substrate degradation during the zincate process. Fig. 2 shows SEM images of samples with or without Cu pretreatment at different zincating time. In these images the substrate surface of Cu-pretreated sample was rapidly and uniformly covered with Zn deposits comparing with the non-Cu-pretreated sample. After 1000 s, Zn particles covered almost completely the Cu-pretreated sample while considerable area of non-Cu-pretreated sample was not covered with Zn deposits, probably due to the low density of the initial nucleation and rather slow deposition rate of Zn for the later sample as confirmed by immersion potential transition. Cu was then electrodeposited on samples prepared with and without Cu pretreatment. SEM observation of Cu electroplated samples showed that the flat and smooth surface of Cu electroplating layer was obtained for Cu-pretreated sample while undulation was observed for the non-Cu-pretreated sample. Defective surface of the Cu electroplating layer were also observed for later sample induced by non-uniform deposition of the Zn layer on the substrate.
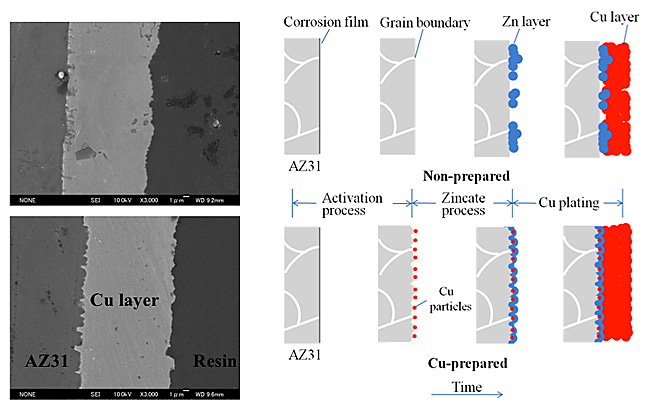
Fig. 3 shows the cross-sectional SEM images of both samples. The defects between the Cu layer and substrate and voids in the Cu layer are observed for non-Cu-pretreated sample. Such defects were induced by the imperfect Zn coverage on the non-Cu-pretreated sample. On the other hand, Cu-pretreated sample shows a smooth and dense Cu surface without defects as confirmed by cross-sectional SEM image. The superior coating was clearly obtained by the preformed dense and uniform Zn layer. An irregular structure of the interface containing pores on the substrate was induced by the acceleration of substrate dissolution due to Cu pretreatment in the activation process. Cu deposition in the pores was confirmed by a cross-sectional SEM image, and this may have contributed to the better adhesion strength of the Cu plating layer, as mentioned below. The coating process for the AZ31 Mg alloy prepared with or without Cu pretreatment is schematically represented in Fig. 3(Right). In the case of the non-prepared sample, the preformed corrosion layer partly dissolved in the activation process and a non-uniform Zn layer was formed on it in the following zincate process. Density of Zn nucleation was low and Zn particles became large compared with those of the Cu-prepared sample. In the Cu electroplating process, Cu was deposited only on the Zn particles with low density, resulting in formation of voids or a defective interface between the Cu layer and the substrate and a defective Cu layer itself with an uneven surface. On the other hand, for the Cu pretreated sample, dense Cu deposits were formed in the activation bath containing a small amount of Cu2+ ions. These Cu deposits acted as catalysts for Zn deposition in the zincate process, and thus a compact Zn layer almost completely covered the substrate. Cu was then electrodeposited on this Zn layer with few defects at the interface and in the film itself. This plating film also showed improvement in adhesion strength.
Pull-off test showed the better adhesion strength of Cu pretreated same (14 MPa) comparing with non-Cu-pretreated sample (9 MPa). Improvement in adhesion strength of Cu pretreated sample was provided by the less defective interface between the coating and substrate and anchor effect as mentioned above.